Regulatory monitoring
in your todo done list
Your regulatory intelligence service : monitors norms, FDA, EU MDR, PMDA, Health Canada, and 100+ other agencies in real time, filtered for your device and markets.
Updated 20 hours ago · See details
Trusted by 500+ customers
THE PROBLEM
Regulatory monitoring does not have to be painful
The information is scattered.
You piece it together from dozens of websites, ChatGPT searches, LinkedIn posts, and colleague tips. You're never sure you haven't missed something.
Cutting through the noise takes forever.
When you do subscribe to official sources, you're buried in updates, and most don't apply to you. Finding what matters takes hours.
Assessing impact and formulating an action plan takes even longer.
Once you find a relevant update, you need to figure out what exactly changed, whether it affects your device portfolio, and what to do about it. That alone can take days.
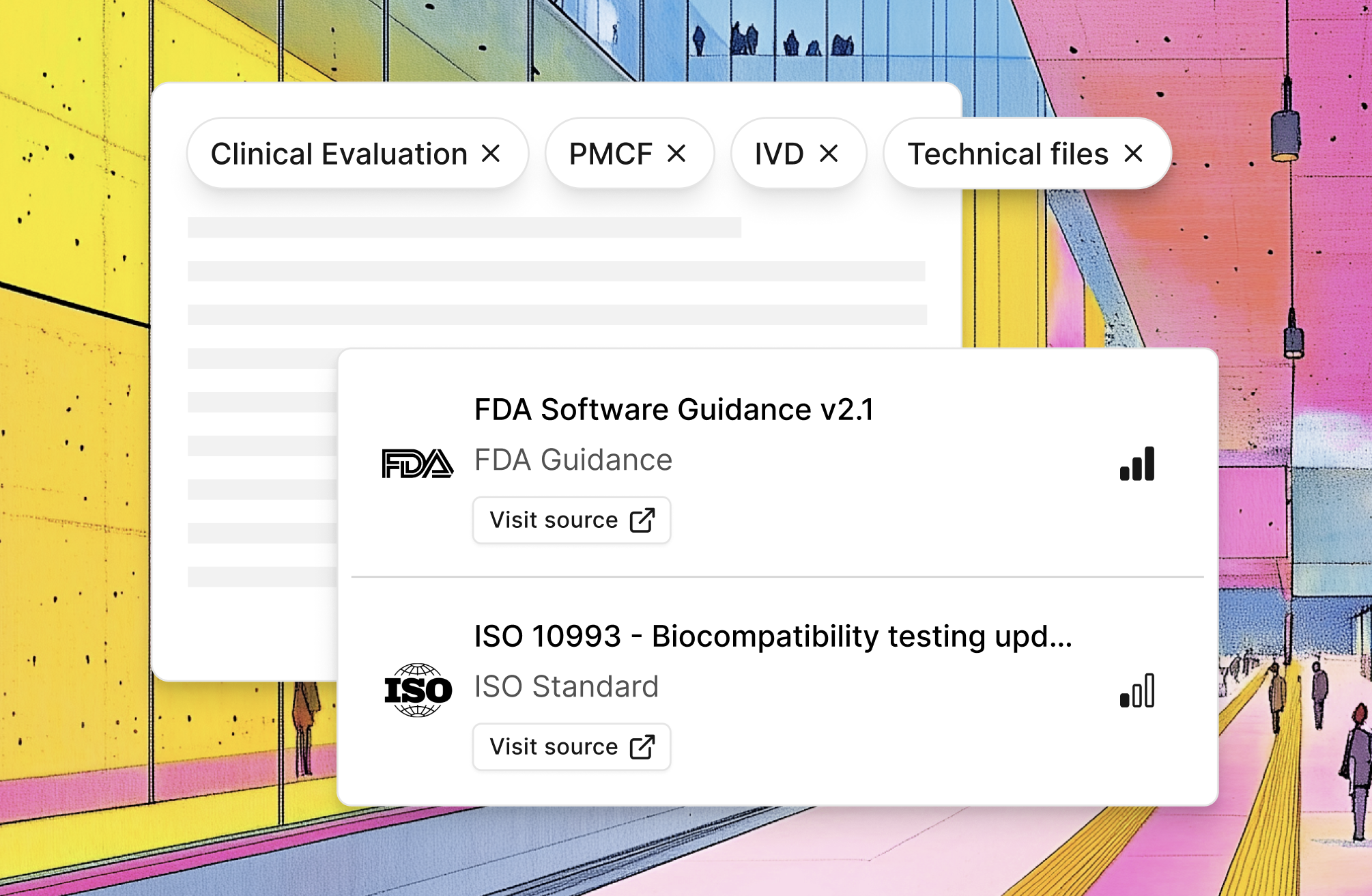
Filtered monitoring you can trust
Filtered by your topics - Clinical Evaluation, PMCF, IVD, Technical Files. Not everything. What you work on.
All official sources - FDA, EU Official Journal, MDCG, ISO, IMDRF, national authorities. Monitored systematically.
Always linked - Direct links to official documents. No hallucinations. Verify in one click.

Impact assessment based on your markets and device portfolio
Redlining built in - See exactly what changed between versions. Additions, deletions, modifications - all highlighted.
Impact assessment - Based on your markets and device portfolio - not just "something changed" but "here's whether it affects you and how urgently."
Action recommendations - Clear next steps for your compliance work and QMS documentation.
CORE PRODUCT
Regulatory Watch
Intelligence on what changed in your regulatory landscape, filtered for your device type and markets. Know exactly what changed and what you need to do.

Want a complete platform tour?
COMPREHENSIVE SOURCES
All the official sources,
all in one place
More than 100 countries supported.










European Commission, FDA, MDCG/MEDDEV, ISO/IEC Standards, IMDRF, US Federal Register, EU NANDO, National Authorities, EMA, EFSA, and more...
HOW IT WORKS
Tailored to your device, your markets, your needs
Generic tools give generic answers. Qalico learns about your specific situation to deliver intelligence that actually matters.
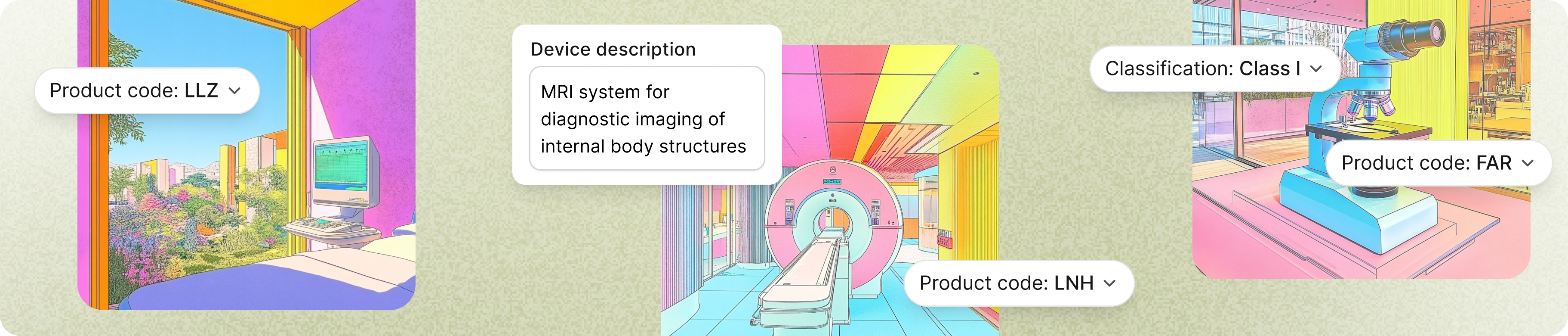
01
Set up your profile
Tell us about your device, product codes, target markets, and clinical claims.
02
We filter for relevance
Our AI uses your profile to search, monitor, and analyze only what applies to you.
03
Get personalized results
No noise — just cited answers, regulatory updates, incident alerts, and documentation tailored to your context.
Be aware of regulation changes before they become a problem or a non-conformity.
Join QARA professionals already using Qalico to save hours, weekly.